Needlestick injury – Community Acquired
Disclaimer
These guidelines have been produced to guide clinical decision making for the medical, nursing and allied health staff of Perth Children’s Hospital. They are not strict protocols, and they do not replace the judgement of a senior clinician. Clinical common-sense should be applied at all times. These clinical guidelines should never be relied on as a substitute for proper assessment with respect to the particular circumstances of each case and the needs of each patient. Clinicians should also consider the local skill level available and their local area policies before following any guideline.
Read the full CAHS clinical disclaimer.
|
Aim
To guide staff with the assessment and management of community acquired needle stick injury.
Background
Community acquired needlestick injuries in children are uncommon but cause a great deal of worry for families. They mostly occur in public places such as the park or street and often after the child intentionally picks up the syringe or needle.
Key points
- The risk of transmission of Blood Borne Viruses (BBV) to a needlestick recipient in a community setting is very low.1
- There are only a few reported cases of members of the public becoming infected by hepatitis B or hepatitis C and none with HIV, following accidental injury from discarded injecting needles in the community setting.1,2
- Hepatitis B transmission risk can be reduced through vaccination and hepatitis B immunoglobulin (HBIG) administration in unimmunised or under-immunised individuals.
- There is no available vaccine or post exposure prophylaxis currently recommended for hepatitis C, though effective treatments are available should acquisition occur.
- Antiretroviral prophylaxis should not be routinely prescribed unless the source of the needlestick is known to be HIV positive. The risk of HIV transmission from community needlestick injury is extremely small (presently no published cases), and antiretrovirals have significant side effects.1
- Follow up is essential.
- Please note that the laboratory is unable to test used syringes for evidence of infective virus under any circumstances.
Estimated risk associated with exposure
| Blood borne virus |
Estimated incidence in WA IV drug users* |
Risk of transmission with a needlestick# |
Calculated maximal risk of transmission✩
|
| HIV |
1.1 - 1.6% |
0.3% |
0 - 0.0048% |
| Hepatitis B |
1.8% |
30% |
0 - 0.54% |
| Hepatitis C |
55 - 58% |
3% |
0 - 1.74% |
* The most likely source of discarded needles in the community
# Figures based on occupational exposure
✩ Calculated from column 1 and 2. Maximal risk is likely to be overestimated.
Assessment
History
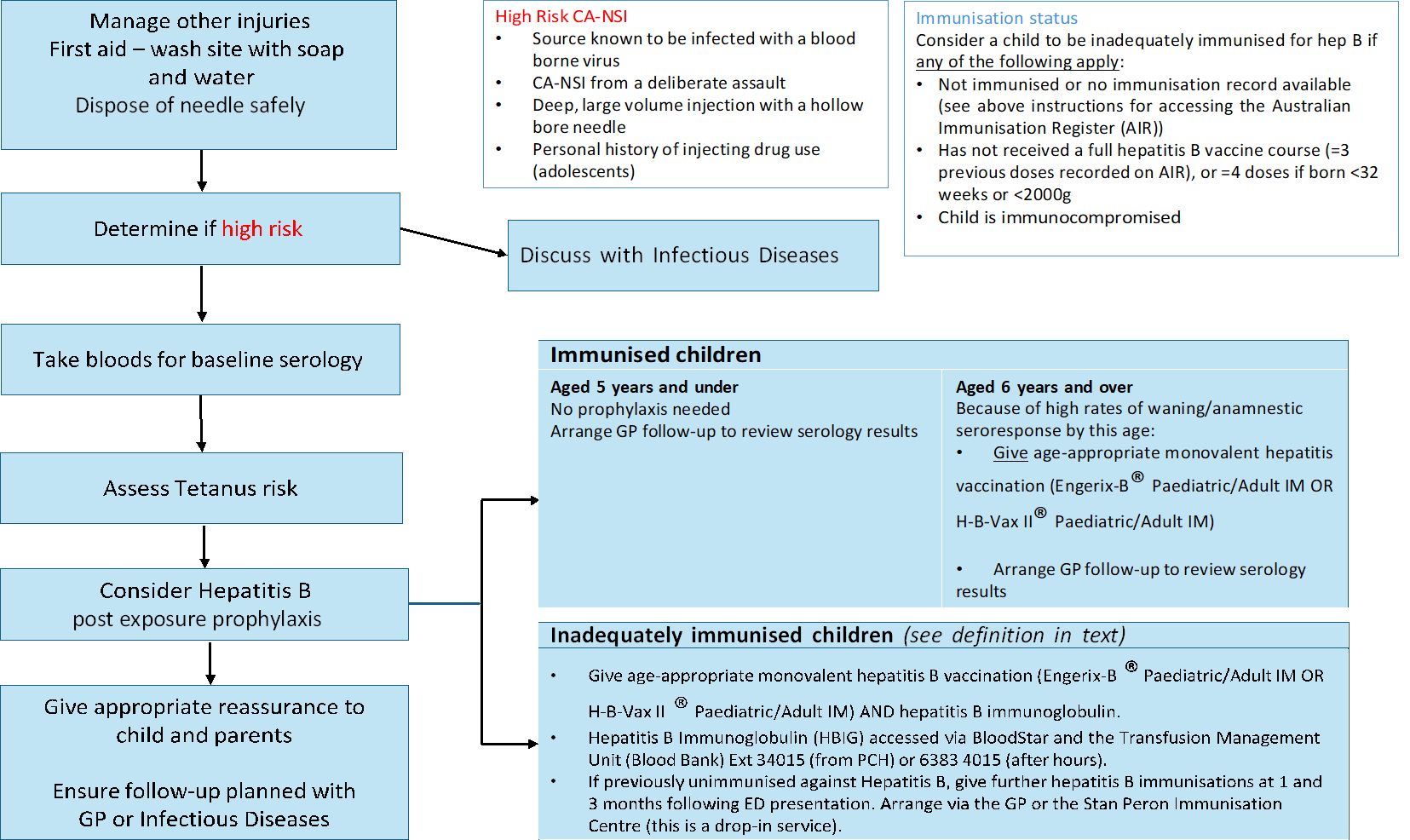
- Assess risk. High risk Community Acquired Needlestick Injury (CA-NSI) is any of the following:
- Source known to be infected with a blood borne virus
- CA-NSI from a deliberate assault
- Deep, large volume injection with a hollow bore needle
- Personal history of injecting drug use (adolescents)
- Assess patient's immunisation status (tetanus, hepatitis B) according to the Australian Immunisation Register (AIR) – this can be accessed through MyHR via i.CM, DMR or via PRODA.
- Further AIR access advice can be provided by the PCH Immunisation Service: Monday to Friday, 8.30am-4.00pm Phone: 08 6456 3721
Investigations
- Take baseline serology (hepatitis B, hepatitis C and HIV). This requires informed verbal consent from parents / carers. Contact microbiology to request urgent processing of serology.
- If the identity of the needle user is known, then the source should also have their blood taken for serology (hepatitis B, hepatitis C and HIV), after obtaining informed consent.
Management
Refer to Appendix 1 - see below.
- Assess and manage associated injuries prior to specific management of CA-NSI
- First aid – wash site with soap and water
- Dispose of needle safely
- If High Risk CA-NSI, discuss with Infectious Diseases
Medical prophylaxis
Tetanus
- Community needlestick injuries are considered tetanus prone wounds. Please refer to Tetanus Prone Wounds and manage according to tetanus vaccination status.
|
Hepatitis B
Unvaccinated / incompletely immunised against Hepatitis B
- If the child fulfills any of the following criteria:
- Not immunised, or no immunisation record available (see above instructions for accessing the Australian Immunisation Register)
- Has not received a full hepatitis B vaccine course (≥ 3 previous doses recorded on AIR), or ≥ 4 doses if born < 32 weeks or < 2000 g.
- Is immunocompromised
- Give: age-appropriate monovalent hepatitis B vaccination (Engerix B® Paediatric/Adult IM OR H-B-Vax II® Paediatric/Adult IM)
AND
Hepatitis B immunoglobulin <30 kg: 100 units IM ≥30 kg: 400 units IM
Access HBIG via BloodStar and the Transfusion Management Unit (Blood Bank) Ext 34015 (from PCH) or 6383 4015 (for urgent requests)
- If previously unimmunised against hepatitis B, give further hepatitis B vaccinations at 1 and 3 months following ED presentation. Arrange via the GP or the Stan Peron Immunisation Centre (this is a drop-in service).
Immunised children
If aged 5 years or younger
- No HBIG or additional hepatitis B vaccinations are required.
If aged 6 years or older
Give age-appropriate monovalent hepatitis B vaccine (Engerix B® Paediatric/Adult IM or H-B Vax II® Paediatric /Adult IM). This is recommended due to high rates of HBsAb <10 mIU/mL in this age group despite immunisations.3,4,5,6
For all patients
Arrange GP follow-up of serology results. Fully immunised patients with a hepatitis B surface antibody (HBsAb) <10 mIU/mL should be offered a booster dose of hepatitis B vaccine and follow-up serology as per Australian Immunisation Handbook advice for hepatitis B non-responders – this can be managed by the patient’s GP.
|
Hepatitis C and HIV
- No post-exposure prophylaxis is available for hepatitis C
- Only children with high-risk CA-NSI are offered HIV post-exposure prophylaxis - discuss with Infectious Diseases
|
Referrals and follow-up
- Needlestick Discharge Information Sheet: standard letter for children with community acquired needlestick injury
- Arrange follow up of baseline Hepatitis B serology with GP as above
- If further Hepatitis B vaccines are required, arrange this course via the GP or Stan Perron Immunisation Centre
- Review at 3 months with repeat testing to ensure no BBV acquisition. This can be with PCH infectious diseases outpatient clinic (complete outpatient eReferral) or with the patient’s GP, according to family preference.
- Follow up serology: HIV, Hepatitis B surface antibody (sAb), Hepatitis B surface antigen (sAg) and Hepatitis C serology to be done at PCH laboratory or other PathWest collection centre approximately 2 weeks prior to the outpatient clinic appointment/GP review. Ensure the patient has
- a pathology request form and
- lidocaine 2.5% with prilocaine 2.5% cream (EMLA®) for the test, if required, which must be prescribed if provided by PCH or it can be obtained from a Community Pharmacy without a prescription. Refer to Supply of Medications by Approved Prescribers (internal WA Health only).
Bibliography
- Papenburg J, Blais D, Moore D, Al-Hosni M, Laferrie`re C, Tapiero B, et al. Pediatric Injuries From Needles Discarded in the Community: Epidemiology and Risk of Seroconversion. PEDIATRICS. 2008;122(2):e487–e92.
- Res S, Bowden FJ. Acute hepatitis B infection following a community-acquired needlestick injury. Journal of Infection. 2011;62:487-9
- Ewe KYH, Blyth CC, McLeod C, Borland M, Bowen AC, Yeoh DK, Campbell AJ. Re-examining Hepatitis B Postexposure Prophylaxis Following Pediatric Community-acquired Needle-stick Injury in an Era of a National Immunization Registry. The Pediatric Infectious Disease Journal 41(1):p 80-84, January 2022.
- Bagheri-Jamebozorgi M, Keshavarz J, Nemati M, Mohammadi-Hossainabad S, Rezayati M, Nejad-Ghaderi M, et al. The persistence of anti-HBs antibody and anamnestic response 20 years after primary vaccination with recombinant hepatitis B vaccine at infancy. Human Vaccines & Immunotherapeutics. 2014;10(12):3731-6.
- Gilca V, De Serres G, Boulianne N, Murphy D, De Wals P, Ouakki M, et al. Antibody persistence and the effect of a booster dose given 5, 10 or 15 years after vaccinating preadolescents with a recombinant hepatitis B vaccine. Vaccine. 2013;31:448-51.
- Poovorawan Y, Chongsrisawat V, Theamboonlers A, Bock H, Leyssen M, Jacquet J. Persistence of antibodies and immune memory to hepatitis B vaccine 20 years after infant vaccination in Thailand. Vaccine. 2010;28(3):730-6.
Appendix 1: Needlestick injury management pathway
Click the image below to download a copy of the needlestick injury management pathway.
| Endorsed by: |
CAHS Drug and Therapeutics Committee |
Date: |
Apr 2026 |
This document can be made available in alternative formats on request for people with disability.